|
Lost two of those electrons and so then it got a positive So then it would not be an ion, it would just be a neutral atom. Originally had 20 electrons and 20 protons. To show that it is a calcium ion, it's likely a situation maybe where the calcium With positive two charge, 20 minus 18 is positive two, and we will denote that with a two plus. So if you wanna know the netĬharge, you take the number of protons, the positive charge and subtract out the number of electrons. Number electrons, I'llĪbbreviate it right over there or I'll shorten it. Protons which provide positive charge is 20. The element is actually how many protons it has, and that's what we have right over here. So why don't you pause this video and see if you canįigure out the charge of that calcium ion. Might be useful to see where calcium sits on that And I will give you a little bit of a tip. Although many of the transition metals can have a wide range of charged ions, some are much more common than others, simply because they are the most stable.Asked, what is the charge of a calcium ion with 18 electrons? So pause this video and see if you can work that on your own. These elements can lose electrons easily, forming different ions. The most common elements to have variable charges are the transition metals. Some elements can have ions with different charges, for example copper can have ions with a +1 or a +2 charge. Some Elements Can Have Multiple Different Ionic ChargesĪlthough it is usually possible to estimate the charge an ion will have based on the number of valence electrons, this is not completely guaranteed. 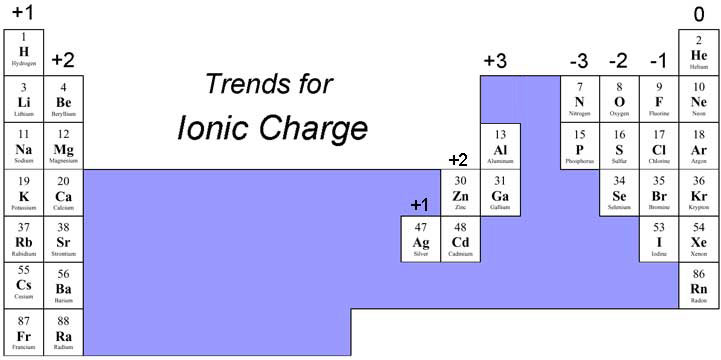 
These have full valence electron shells, meaning that they do not form ions. They have accepted an electron in order to fill their valence electron shells and, therefore, have more electrons than they do protons.Įlements in group 8 are the noble gases. This pattern continues, meaning that elements in group 7 have ions with a charge of -1. This is because the elements in group 1 have one valence electron and those in group 2 have two valence electrons. Elements in group 2 will have a charge of +2. Elements in group 1 (that is the first column of the periodic table) will usually have ions which have a +1 charge. It is possible to estimate the charge that an ion will have from the position the element occupies in the periodic table of elements. The Periodic Table of Elements Provides Information on Ionic Charges If the atom has to lose three electrons, it will have a charge of +3. If the atom has lost one electron, it has a positive charge (because it has more positively charged protons than it does negatively charged electrons) and thus is can be thought of as having a +1 charge. The number of electrons it will take to fill the valence electron shell is known as the ‘charge’ of an elemental ion. If a valance electron shell is emptied, it ceases to exist (from a chemistry perspective) and the next shell in becomes the valence shell. This will have filled (or emptied) an outer valance electron shell. If an element is in ionic form, it has donated (given up) or accepted electrons. 
Some elements in the periodic table of elements are stable, in that they have full valence electron shells and are, therefore chemically inert. When a substance has a full outer electron shell (this shell is known as the valence shell), it becomes stable and is unlikely to react. Although atoms are electrically stable in their base form, where they have the same number of protons and electrons, atoms will also react to form complete electron shells. Electron Shells and IonsĮlectron shells are areas around the nucleus of an atom which are occupied by electrons. Those which are positively charged are known as cations, and those with a negative charge are anions. Ions are atoms that carry an electrical charge. Ions are formed when an atom (which was previously electrically neutral) either gives up some of its electrons, or gains some electrons from other atoms. This balance in the number of protons and electrons means that the atoms are balanced, and therefore carry almost no electrical charge.Īlmost all of the elements in the periodic table of elements are capable for forming ions, however, and their position on the periodic table allows scientist to predict how they will do this. Because the periodic table of elements is only looking at single elements, and not compounds, it is based on when the element is not in an ionic form, meaning that it has the same number of electrons as protons. The organisation of the periodic table is based on the structure and nature of atoms of each, individual single element. The periodic table is a way of categorising elements in order to help scientists and researchers.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
